Vesicular Stomatitis Virus
Also Known As – VS
Caused By – Vesicular Stomatitis Virus – VSV – Alagoas Virus – Brazil Virus – Cocal Virus – Piry Virus – Chandipura Virus – Isfahan Virus
Introduction
Vesicular Stomatitis is an oral disease of cattle, horses, pigs and people, caused by a rhabdovirus, of which there are two distinct serotypes: VSV-New Jersey (VS-NJ) and VSV-Indiana (VS-IN). Although antibodies have been found in many species of mammal and bird, no clinical signs appear to develop.
The main importance of VS is its identical presentation to Foot and Mouth Disease, in fact they cannot be clinically differentiated.
VS is zoonotic and can cause acute flu-like disease in humans.
This disease is notifiable to the World Organisation for Animal Health (OIE)
Distribution
VS is present exclusively in the Western hemisphere, mainly in the Americas, including parts of the Caribbean and South America.
VS is transmitted by a range of insect vectors including the house fly and Culicoides midge.
The virus can also be transmitted via direct contact with lesions, saliva and fomites.
Signalment
Young animals <1 year old are less likely to exhibit clinical signs although seroconversion occurs.
Disease occurs seasonally in the late Summer and Autumn in endemic countries.
Clinical Signs
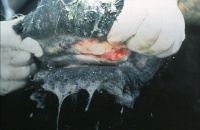
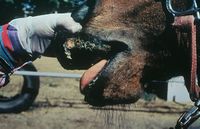
VS is characterised by its appearance – beginning as blanched macules and developing into vesicular lesions on the tongue, oral and nasal mucosa, mammary glands, external genitalia and coronary band. When the vesicles rupture after 24-48h, erosions and excoriations remain in the affected mucosa. Hypersalivation often also occurs. Coronary band lesions cause lameness and defective horn growth.
Other clinical manifestations include depression, weight loss, low productivity, agalactia and mastitis. Animals with genital lesions may be reluctant to breed.
Affected animals are often pyrexic, anorexic and dysphagic due to reduced food intake and interference of lesions with prehension/pain when eating.
Shedding ceases 6-7 days post-infection [1] and lesions resolve within 14 days.
Once recovered, animals do not maintain infection or shed the virus.
Deaths from VS are rare and usually due to secondary complications.
Diagnosis
Definitive diagnosis requires laboratory testing. Virus isolation can be attempted from nasal/oral swabs, epithelial tags, skin scrapings or biopsies. Only active lesions will contain infective virus particles. PCR is potentially more sensitive but not widely used.
Serlogical tests are routinely used and will also differentiate serotype. Competitive ELISA (cELISA) is used as a screening test and Serum Neutralisation (SN) and Complement Fixation (CF) are also commonly used to diagnose VS. Tests that detect IgG such as the cELISA and SN may remain positive for up to 3 years after infection.
CF is more indicative of recent infection, especially when rising titres are present.
On histopathology, intercellular oedema is present in the Malphigian cell layer and epithelial cell necrosis is evident. The necrotic cells are strongly eosinophilic and nuclei are pyknotic. Basal cells are separated by vesicular fluid. Other non-specific inflammatory changes are usually also present.
Treatment
Disease is self-limiting and usually requires no treatment. Secondary bacterial infection can delay healing of lesions and usually respond to antibiotics. Symptomatic care may be indicated in severe cases.
Control
Several vaccines are being developed or used experimentally but none are commercially available in the USA.
Appropriate biosecurity, disinfection protocols, temporary isolation of infected animals/groups and limiting exposure to insect vectors may help to reduce the scale of outbreaks.
Quarantine procedures are in place in many countries when VS is confirmed and these should be adhered to and relevant bodies informed.
References
- ↑ Katz, J. B., Eernisse, K. A., Landgraf, J. G., Schmitt, B. J. (1997) Comparative performance of four serodiagnostic procedures for detecting bovine and equine vesicular stomatitis virus antibodies. J Vet Diagnostic Investigation, 9(3):329-331; 5
Animal Health & Production Compendium, Vesicular Stomatitis datasheet, accessed 06/06/2011 @ http://www.cabi.org/ahpc/
Animal Health & Production Compendium, Vesicular Stomatitis virus datasheet, accessed 06/06/2011 @ http://www.cabi.org/ahpc/