Complement Associated Diseases
Complement
- Complement is so called because it complements the function of antibody.
- Complement is a triggered enzyme cascade.
- These are a group of plasma systems.
- Other examples include:
- The clotting system.
- The kinin cascade.
- The triggered enzyme cascades are effector mechanisms that can produce a rapid and amplified response to a trigger stimulus.
- Have adaptive value if appropriately invoked.
- May be harmful if triggered inappropriately.
- The reaction pathways and control mechanisms involved are complex.
- There are more than 20 different proteins ) in the complement cascades.
- Most are enzymes or pro-enzymes.
- The complememnt system evoluntionarily pre-dates the immune system.
- Has evolved as one of the main innate protective mechanisms of invertebrates.
- Complement works largely by triggering acute inflammation and by promoting phagocytosis by macrophages and neutrophils.
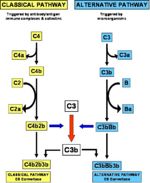
Complement Fixation
- The central event in complement activation is the hydrolysis of the major complement protein, C3.
- C3 is a large 2-chain molecule.
- Hydrolysis results in the removal of a small peptide, C3a (which itself is pro-inflammatory).
- This leaves C3b.
- C3b is highly reactive but very short-lived.
- This leaves C3b.
- The hydrolysis of C3 occurs spontaneously and slowly in plasma.
- The reactive C3b formed is rapidly inactivated.
- Some substances can interact to alter aspects of the complement system.
- Bacterial cell surfaces.
- Protect the reactive C3b from rapid inactivation.
- It is still inactivated, but more slowly.
- This is the basis of alternative pathway of complement activation.
- This is the archaic complement system.
- Protect the reactive C3b from rapid inactivation.
- Antibody-antigen complexes
- The immune system has optimized complement activation via antibody-antigen complexes.
- This is termed the classical pathway of complement activation.
- Bacterial cell surfaces.
- The difference between the alternative and classical pathways is in the mechanisms of C3 breakdown to C3b.
- Once C3 is hydrolysed, the terminal pathway is the same for both mechanisms.
- Often called the membrane attack complex (MAC).
- Involves the binding of C5-C9.
- Once C3 is hydrolysed, the terminal pathway is the same for both mechanisms.
Alternative Pathway Activation
- Control proteins in plasma are responsible for the rapid inactivation of C3b to iC3b.
- These are known as Factors I and H.
- C3b has a binding site that anchors it to any surface.
- The surfaces of certain activators therefore stabilise the usually short-lived C3b in the absence of antibody.
- Gram-negative bacteria, yeasts and fungi are the most efficient activators.
- The surfaces of certain activators therefore stabilise the usually short-lived C3b in the absence of antibody.
- When active C3b is bound to particle surfaces, it is protected from inactivation by another complement component, properdin.
- C3b can then bind Factor B.
- This produces the complex C3bB.
- C3b can then bind Factor B.
- C3bB is the only substrate for a plasma enzyme known as Factor D.
- Factor D splits a small peptide (the Ba peptide) from Factor B.
- This splitting generates C3b¯Bb.
- An active enzyme that splits C3.
- The bar indicates an active enzyme.
- This splitting generates C3b¯Bb.
- Factor D splits a small peptide (the Ba peptide) from Factor B.
- C3b¯Bb acts on C3 to generate more C3b.
- C3b then generates more C3b¯Bb.
- An efficient positive feedback loop.
- C3b then generates more C3b¯Bb.
- C3b¯Bb can also bind C3b to form C3bB¯b3b.
- C3bB¯b3b is one of the two enzymes that activates the Membrane Attack Complex (MAC).
- C3bB¯b3b splits C5 into:
- C5a, a small pro-inflammatory peptide.
- C5b, the initiator of the MAC.
- C3bB¯b3b splits C5 into:
- C3bB¯b3b is one of the two enzymes that activates the Membrane Attack Complex (MAC).
- At the same time as C3bB¯b3b is being formed, Factors I and H are acting to breakdown C3b to iC3b.
- The C3b being broken down may be:
- As single C3b or in the complex C3b¯Bb.
- In plasma or on bacterial surfaces.
- iC3b is inactive in the complement cascade but is a major target for phagocytes.
- Phagocytes have large numbers of iC3b receptors (complement receptors) which give opsonization when engaged.
- The C3b being broken down may be:
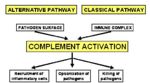
- The main effects of alternative complement activation are therefore:
- To coat bacteria with iC3b.
- A major target for phagocytosis by macrophages and neutrophils via the complement receptors.
- To induce an acute inflammatory response via C3a and C5a.
- Chemotactic for neutrophils .
- Induce the production of the cytokines (IL-1β and TNFα) responsible for acute inflammation.
- To coat bacteria with iC3b.
Classical Pathway Activation
- There are two triggers for the classical pathway of complement activation.
The binding of antibody to antigen
- Only IgM and certain IgG subclasses can activate complement this way.
- Immune complexes trigger complement activation by binding C1.
- C1 cross-links two antibody molecules.
- C1 is a complex of:
- C1q
- Looks like a bunch of 6 tulips.
- Each "flower" consists of a globular protein head and a collagen "stem".
- At least two C1q globular heads must bind to antibody before the complement cascade is triggered.
- C1r
- C1s.
- C1q
- C1r and C1s become activated when at least 2 C1q heads are antibopdy-bound.
- Form the enzyme C1 esterase.
- C1 esterase first digests C4 to C4a and C4b.
- C4b binds to the antigen.
- C4b then binds C2.
- C2 is digested by C1 esterase to form C2a and C2b.
- C4b and C2b complex to form the enzyme C2b¯4b.
- C2 is digested by C1 esterase to form C2a and C2b.
- C2b¯4b digests of C3 into C3a and C3b.
- The production of C3b can now be amplified by the same mechanism as the alternative pathway.
- The binding of one C1q molecule produces one C1 esterase molecule that then cause the binding of many hundreds of C4b¯2b molecules.
- The C3b molecule that is produced by the action of this enzyme can also bind to it.
- Forms C4b2¯b3b.
- This activates C5 and initiates the MAC.
- Forms C4b2¯b3b.
- The C3b molecule that is produced by the action of this enzyme can also bind to it.
- Like the alternative pathway, another major effect of classical pathway activation is to produce iC3b due to the actions of Factors I and H.
- Promotes phagocytosis and initiates inflammation.
- All the Ca fragments (C2a, C3a, C4a, C5a) are chemotactic for neutrophils and induce of acute inflammation.
The binding of soluble lectins to pathogens
- E.g. collectins.
- Lectins are proteins that bind carbohydrates.
- In this case, they bind carbohydrates that have a terminal mannose residue.
- They are called mannose-binding lectins.
- Are secreted by the liver into plasma.
- In this case, they bind carbohydrates that have a terminal mannose residue.
- The action of lectin binding to carbohydrate activates plasma-associated proteases called mannose-binding lectin associated proteases (MASPs).
- These act on C4 and C2 in the same way as C1 esterase.
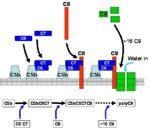
Membrane Attack Complex
- This is the lytic pathway of complement function.
- Can be initiated via either C3bB¯b3b or C4b2¯b3b.
- Split C5 to:
- C5a - pro-inflammatory.
- C5b - the first molecule of the MAC.
- Split C5 to:
- C5b is very short-lived and biologically active.
- Rapidly attaches to cell surfaces and binds to one C6 molecule.
- The C5bC6 complex binds one C7 and then one C8 molecule in turn.
- Finally, about 16 C9 molecules bind and polymerize within the cell membrane.
- This polymerization of C9 results in a small pore being formed.
- Causes cell lysis by osmotic shock.
- This polymerization of C9 results in a small pore being formed.
Biological Activities of Complement Components
Opsonisation
- Once the complement system has been triggered it deposits a shell of protein on the bacterial cell surface.
- C4b, C3b, C5b and C7 molecules contain active binding sites that anchor the complex to the surface.
- The major protein on the cell surface is iC3b.
- As well as some of the smaller C3 breakdown products (e.g. C3d), iC3b act as a target for phagocytosis.
- There are very avid receptors on phagocyte membranes for these complement fragments.
- As well as some of the smaller C3 breakdown products (e.g. C3d), iC3b act as a target for phagocytosis.
- Complement-mediated opsonization of microorganisms is several thousand times more efficient that innate receptors.
- The complement fragments released after complement activation are chemotactic for phagocytes.
- C2a C3a, C4a and especially C5a.
Inflammation
- The smaller complement peptides are very efficient at inducing inflammation.
- C3a and C5a.
- They attract granulocytes to the site of complement activation.
- Also stimulate their degranulation.
Cell Lysis
- Finally, the later components (C5 – C9) can kill pathogens directly by causing cell lysis.
- This is effective against encapsulated bacterial infection like Neisseria and Meningococci.
COMPLEMENT INHIBITORS
As mentioned above complement is a very powerful system that can be triggered by only small stimuli. Inappropriate activation can, therefore be harmful. Consequently a range of control mechanisms has evolved to control this. Decay accelerating factor (DAF) is present on cell membranes as well as a secreted product; it hastens the degradation of C1 esterase and controls the Classical Pathway. Factors I and H function to breakdown C3b hence controlling positive feedback by inhibiting C and preventing the complement cascade from running to exhaustion each time it is activated. Complement receptor 1 (CR1) is present on many cell types especially RBCs; it functions to bind C3d - the breakdown product of C3b – resulting from the action of Factors I and H. On it binds potentially inflammatory immune complexes in plasma; these are then transported to the liver where they are phagocytosed by the hepatic macrophages and removed. A common inflammatory disease resulting from poorly eliminated immune complexes is globerulonephritis. Finally there is the other membrane-associated inhibitor - CD59. This binds the first molecule of C9 when it inserts into a cell membrane. This prevents the polymerisation of C9 obviating pore formation and cell lysis. It acts as a protective mechanism for the host cells.
COMPLEMENT ASSOCIATED DISEASES
Most diseases associated with complement are linked to deficiencies of certain components. Deficiencies of components like C1, C2, C4, CR1 and DAF lead to autoimmune disease resulting from the inefficient removal immune complexes. This causes these complexes to lodge in many of the small capillary beds and induce inflammation. Most commonly this brings about such diseases as glomerulonephritis, vasculitis, rheumatoid arthritis and inflammation/irritation of the skin, for instance Systemic Lupus Erythematosis.
Figure 3 Deficiencies of C3 and Factor B as well as the inhibitors Factors H and I lead to chronic infection. Lack of the inhibitors results in the exhaustion of the supply of C3 causing functional deficiency of C3. Without C3 both the lytic pathway and much more importantly, opsonization are not optimally functioning. Lack of C6 results in very few effects other that the inability to eliminate encapsulated bacterial infections (e.g. Neisseria spp). The rare genetic deficiency of C1 esterase inhibitor (an enzyme which controls the functioning of C1 esterase) results in hereditary angiodæma. Lack of this enzyme causes inappropriate activation of C2 and the production of large quantities of C2a (also known as C2 kinin). This is a potent inducer of inflammation and of vasodilatation. The vasodilatation results in the oedema.