Bovine Viral Diarrhoea Virus
Also known as: Bovine Viral Diarrhoea — Bovine Virus Diarrhoea (Virus) — Mucosal Disease
Introduction
Bovine viral diarrhoea is a viral disease that affects cattle worldwide. Caused by a Pestivirus, it gives rise to significant economic losses in both dairy and beef cattle through its effects on production and reproduction. Bovine viral diarrhoea virus can lead to a variety of clinical outcomes that range from subclinical infections to the more severe presentations including abortion, infertility, and the fatal mucosal disease. The condition is highly immuno-suppressive and secondary respiratory and enteric complications often occur.
Bovine Viral Diarrhoea Virus
Classification
The viral aetiology of BVD was first established over 60 years ago, but it was not until the 1960s that the agent was assigned to the newly penned "Pestivirus" genus. At this stage Pestiviruses were considered to be non-arthropod-borne Togaviruses; later, sequencing of genomic RNA showed that they are taxonomically better suited to the Flaviviridae family[1][2]. Many members of the Flaviviridae family are indeed arthropod-borne, such as the Flaviviruses West Nile Virus and Yellow Fever Virus. However, Pestiviruses are not transmitted by insects, and the genus includes pathogens of cattle (BVDV), sheep (Border Disease Virus) and pigs (Classical Swine Fever Virus).
Virus Structure
The BVDV genome comprises a single strand of positive sense RNA which is around 12.3 kilobases in length[3]. The genome is read in one 3898-codon open reading frame that contains no non-coding sequences. BVDV polyprotein is translated directly from the ORF and is cleaved by viral and cellular proteinases to form mature viral proteins[3][4]. At either end of the ORF, 5’ and 3’ untranslated regions exist. These regions are long, allowing them to accomomdate functions conferred in eukaryotic DNA by the 5’ cap and the 3' poly-A tail, such as controlling the initiation of translation, facilitating the entry of replicases, and contributing to RNA stability[4]. BVDV's RNA genome encodes both structural and non-structural proteins. These include Npro, whose protease action generates the N-terminus of the protein C. C is the capsid protein that packages genomic RNA and assists in the formation of the eventual enveloped virion. Erns, E1 and E2 are all glycoproteins, with Erns possessing RNase activity involved in viral replication and pathogenesis[5]. E1 is membrane-anchored and initiates the translocation of the antigenic protein E2 to the envelope[3]. P7 has an uncertain function[6]. NS2-3 is the first non-structural protein to be translated. Sequence similarities are shown by NS2-3 to a region that in other Flaviviridae is split into two distinct polypeptides, NS2 and NS3. In BVDV, NS2 and NS3 can be expressed as separate polypeptides: NS3 is found exclusively in cytopathic isolates from 6 hours post-infection, making it a marker of this biotype[3]. NS2 is also expressed as a discrete polypeptide in some cytopathic isolates. NS2-3, along with the other non-structural proteins, plays an important role in genome replication. A serine protease domain within NS2-3 functions to release NS4A, NS4B, NS5A and NS5B[7]. NS4A is a cofactor for the serine protease[7], and NS5B possesses an RNA-dependent RNA polymerase activity[8]. Knowledge of the role of NS4B is limited.
Newly formed genomic material is packaged by structural proteins to create the BVDV virion which is 40-60nm in diameter. The capsid is surrounded by a membranous envelope, in which the glycoproteins E1 and E2 are anchored. Naked BVDV RNA is infectious[3][4], and so it can be deduced that the virions do not contain enzymes necessary for RNA replication: these are provided by the host cell.
Virus Genotypes
There are two antigenically distinct[9] genotypes of BVDV, type 1 and type 2, which are most accurately characterised based on sequence variation. BVDV-1 and BVDV-2 contain 11 and 3 subtypes respectively, which have been demonstrated by analysis of the 5’UTR[10]. Despite the large antigenic differences between the genotypes, some cross-protection against type 2 viruses is afforded by type 1 vaccines[11][12][13].
In general, the genotypes do not differ in virulence. It was, at one time, considered that BVDV-1 species which are found worldwide caused milder disease [14], whereas BVDV-2 isolates typically caused more severe disease which is often haemorrhagic and was associated with a high mortality rate[11][15]. This is no longer the case. The relationship between genotype and virulence is, however, not fixed: some type 2 strains cause mild or subclinical disease[16], and the spectrum disease caused by type 1 viruses is broad. Type 2 viruses were first reported in Canada and the USA and have a more limited distribution than type 1 isolates.
Virus Biotypes
BVDV isolates from either genotype can be of a cytopathic or non-cytopathic biotype. Non-cytopathic (ncp) viruses produce no visible effects in cell culture, whereas infection with cytopathic (cp) viruses gives cell vacuolation and death. Although non-cytopathic isolates are responsible for the majority of BVDV infections worldwide, cytopathogenicity gives no indication of disease-causing potential. Cytopathic biotypes of bovine viral diarrhoea virus are always isolated alongside non-cytopathic strains, and are found in cases of mucosal disease, a fatal BVD-associated condition[17].
Cytopathic viruses originate from non-cytopathic strains by mutation, including viral gene rearrangements, duplications and deletions[18], and insertions of cellular origin, such as ubiquitin sequences. Point mutations in the NS2 region and various RNA recombination events are also important[19]. Serologically, the two BVDV biotypes are indistinguishable, but on a molecular level cytopathic viruses produce an additional protein, NS3, not found in cells infected with non-cytopathic virus[20][21][22]. This marker molecule arises from when NS2-3, expressed in non-cytopathic isolates, is cleaved at a site created by the mutations above[23].
Transmission and Epidemiology
In most countries, BVDV is endemic and studies detecting antibody have estimated that between 70 and 100% of herds are either currently infected or have previously been infected with bovine viral diarrhoea virus[24].
BVDV can be transmitted from infected to susceptible cattle in several ways. Firstly, direct contact with an animal shedding BVDV in its secretions and excretions can cause disease. Virus is shed by both acutely and persistently infected (PI) animals but levels of shedding are much higher in persistently infected cattle, which are the natural reservoir for virus. It is estimated that the incidence of persistently infected animals is 1-2% of cattle less than one year of age and may be higher in infected herds. On a farm, PI cattle are often found in cohorts of similarly aged animals. This is because persistent infections arise when pregnant animals are acutely infected in early pregnancy, and so an outbreak of acute, possibly subclinical, BVD in pregnant cattle can later result in a "batch" of PI calves.
Transmission to heifers and cows may also occur venereally, via artificial insemination or during embryo transfer, as acutely and persistently infected bulls shed bovine viral diarrhoea virus in their semen[25]. The testes is an immunoprivileged site, and the virus can persist in this location despite otherwise systemic clearance[26]. Indirect spread is possible: BVDV has been shown to spread through the re-use of needles, nose tongs[27] and rectal gloves[28], and blood feeding flies also give transmission.
Pathogenesis
Following entry and contact with the mucosa of the oral or nasal cavities or the reproductive tract, BVDV replicates in epithelial cells and has a predilection for the palatine tonsil and the nasal mucosa. From here, the virus spreads to regional lymph nodes before a viraemia becomes established. Virus can be disseminated free in the blood, or associated with leukocytes, particularly lymphocytes and monocytes[29]. Bovine viral diarrhoea virus can then gain access to many tissues, but shows a preference for lymphoid tissue, reaching its highest concentrations in the tonsil, thymus and ileum. Bone marrow[30] and intestinal mucosa are often infected, and the lymphoid tissue of the Peyer's patches is frequently depleted. However, there is variation between strains as to which tissues are specifically infected and in general, a wider distribution is associated with higher virulence.
Diagnosis
Clinical appearance of BVD infection is highly variable, often underlying many calf infections due to its immunosuppressive effect permitting secondary respiratory and enteric infection. Furthermore, the persistently infected calves can appear either unthrifty or perfectly normal. Therefore laboratory testing for the virus is essential.
Clinical Signs
It might be expected from the nomenclature (bovine viral diarrhoea virus) that diarrhoea is a key clinical feature in BVDV infection, but it is not a major clinical sign. The clinical presentation can actually manifest in a variety of ways ranging from subclinical disease to the fatal muscosal disease[31]. Virulence factors related to genotype and strain are partially responsible for these variations, but host factors are also important. Pregnancy status, stage of gestation, immunity and the level of development of the foetal immune system all contribute to the outcome of BVDV infection.
Acute Infections: Non-Pregnant Cattle
In the naive, non-pregnant, immunocompetent animal, BVD is normally mild: it is estimated that 70 to 90% of BVDV infections cause no obvious clinical signs[32]. If these subclinically affected cattle are observed closely, body temperature may marginally rise and mild leukopenia and agalactia may be seen [33][34]. When clinical disease does occur in these animals, morbidity is high amongst cattle of 6-12 months of age. Following a 5-7 day incubation period, pyrexia and leukopenia is seen. Viraemia arises on days 4-5 days post-infection, and continues until around day 15[35]. Although some cattle suffer diarrhoea in BVDV infection, the disease no longer seems to present as herd outbreaks of diarrhoea[31]. Clinical signs more commonly include depression, anorexia, occulo-nasal discharge, decreased milk production and oral lesions[36], with a rapid respiratory rate resembling pneumonia sometimes apparent[33]. Acutely infected, non-pregnant animals shed low concentrations of virus compared to persistently infected cattle[35], and antibodies are produced 2-4 weeks post-infection which persist for many years[36].
Acute BVDV infection causes a significant leukopenia, hampering the host's defences against invading pathogens. This BVDV-associated immunosupression has a particularly important role in bovine respiratory disease: an association has been demonstrated between BVDV antibody titre and treatment for respiratory disease[37]. BVDV is the virus most frequently isolated from pneumonic lungs and is often found in association with Pasteurella haemolytica[36], causing severe fibrino-purulent bronchopneumonia and increasing the total lesion area by 35-60% compared to pasteurellosis alone[31]. Synergism is also displayed with parainfluenza, bovine rhinotracheitis and respiratory syncitial viruses.
BVDV infections in naive, non-pregnant animals are usually mild; however outbreaks of a severe form of BVD became evident in the late 1980s in the USA and Canada[11][38]. These were characterised by the acute onset of diarrhoea, pyrexia and milk drop, with some cases proving fatal. These oubtreaks were associated with genotype 2 viruses, and it transpired vaccination with type 1 vaccines had afforded poor cross-protection in these instances due to non-compliance with instructions. Initially, BVDV-2 infection was seen less frequently than disease related to type 1 virus, but was associated with a haemorrhagic syndrome. The haemorrhagic syndrome is characterised by severe thrombocytopaenia leading to haematochezia, petechiation and epistaxis[39] and has now been described in both Europe and North America. Severe disease is also possible with virulent type 1 infection, presenting as high fever, oral ulcerations, eruptive lesions of the coronary band and interdigital cleft, diarrhoea, dehydration, leukopenia, and thrombocytopenia. Thrombocytopenia may give petechiation of the conjunctiva, sclera, nictitating membrane and the mucosal surfaces of the mouth and vulva, as well as prolonged bleeding from injection sites[40].
Acute Infections: Pregnant Animals
When acute BVDV infection occurs during pregnancy, the dam may show any of the clinical manifestations that are seen in non-pregnant animals. BVDV is able to cross the placenta and infect the developing foetus and so there may be additional outcomes of infection that depend on the stage of gestation. If infection becomes established at the time of insemination, conception rates may be reduced, and early embryonic death is increased when the virus is introduced at a slightly later stage[41][42]. Foetal infection in the first trimester (50-100 days) may also result in death, although expulsion of the foetus often does not occur until several months later. An additional effect of foetal infection before 120 days gestation is the birth of persistently infected (PI) calves.
Congenital defects can arise from transplacental infection between days 100 and 150. This is caused by an inappropriate inflammatory response mounted to BVDV by the immune system, which is undergoing the final phase of development at this stage[35]. Examples of common congenital abnormalities include defects of the thymus, ocular changes and cerebellar hypoplasia[31]. Calves with cerebellar hypoplasia are ataxic, reluctant to stand and may suffer tremors[36], and ocular pathology often causes blindness and cataracts. Localisation of virus to the vascular endothelium gives vasculitis, leading to oedema, hypoxia and cellular degeneration. Weak, stunted calves may also be produced by BVDV infection in the second trimester.
Infection in the third trimester trimester (over 180-200 days) elicits a response from the fully-developed immune system, giving rise to normal but seropositive calves.
Persistent Infections
Foetal infection with a non-cytopathic BVDV virus before 120 days gestation may result in the birth of calves persistently infected with and tolerant to bovine viral diarrhoea virus. At this stage in gestation, the immune system is partially competent and recognises the BVDV antigen as self, meaning that no response is mounted. The calf therefore becomes tolerant to the virus which persists into neonatal life[17]. Persistently infected animals can be identified at birth as being antigen-positive but seronegative. However, colostral transfer of maternal immunity or infection with a heterologous strain of BVDV can make these animals seropostitive, so care must be taken when timing and interpreting tests.[43]
Persistently infected animals continuously shed large amounts of virus throughout their lives, providing a major source of infection for naive cattle[24]. Persistently infected dams produce persistently infected calves, resulting in family lines capable of maintaining the virus in a herd[36]. It is estimated that 1-2% of the cattle population and up to 13% of foetal calves are persistently infected[31].
50% of persistently infected cattle die within the first year of life[35]. Animals may be undersized and slow-growing, and are predisposed to other diseases.[44] Persistent infection with BVDV is the prerequisite for developing mucosal disease[17].
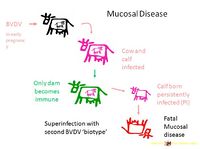
Mucosal Disease
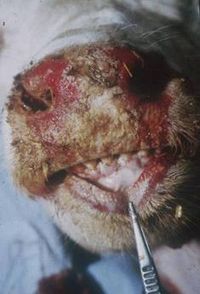
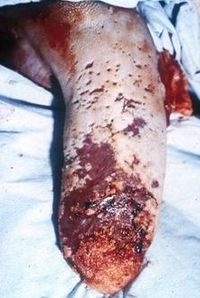
Mucosal disease is an invariably fatal condition of 6-18 month-old cattle[45]. Disease follows a course of several days to weeks and intially presents as pyrexia, depression and weakness. Anorexia leads to emaciation, and animals suffer watery, foul-smelling and sometimes bloody diarrhoea. Dehydration ensues. As suggested by the name, lesions are localised to mucosal surfaces. These include the oral mucosa, tongue, external nares, nasal cavities and conjunctiva[31], where large lesions cause excessive salivation, lacrimation, and oculo-nasal discharge. The coronet and interdigital surface are also affected, causing the animal to become disinclined to walk and eventually recumbent.
Mucosal disease arises from superinfection of persistently infected animals with a cytopathic virus antigenically similar to the original, non-cytopathic strain persisting in the animal. In one animal, a cytopathic virus is produced by mutation of the persistent non-cytopathic virus. The new cytopathic isolate can then be transmitted to other animals where it will cause mucosal disease if they are persistently infected with the same non-cytopathic strain. Immune tolerance induced by the persistent virus prevents the immune system recognising the superinfecting cytopathic strain: the two biotypes are said to be "homologous" to the immunotolerance.[46]. "Heterologous" superinfection with a non-related cytopathic biotype does not result in mucosal disease because a normal immune response is mounted.
Laboratory Tests
There are several techniques available for the laboratory diagnosis of BVD. These can detect antibody to BVDV or parts of the virus itself.
Tests that detect anti-BVDV antibody include the serum neutralisation test, and an ELISA[31]. The serum neutralisation test depends on the ability of antibodies in the serum to neutralise BVD virus and thereby prevent infection of cell culture. The test takes four to seven days and requires cell culture facilities and an experienced observer. The ELISA can detect either BVDV antibody or BVDV antigen. The BVDV ELISA test can be completed within hours and is simple to perform. Because antibody against BVDV is prevalent in most cattle populations, a single serologic test is not usually sufficient for diagnosis of a recent infection. Therefore, an increase in antibody titre between paired serum samples must be more than four-fold to confirm recent infection[40].
Viral antigen or RNA can be detected using clinical specimens or tissue samples. Bovine viral diarrhoea virus can be isolated from blood, nasal swabs or tissues to confirm active infection, and demonstration of virus in samples obtained at least three weeks apart is suggestive of persistent infection. The best tissues for virus isolation are skin, spleen, lymph node and segments of the gastrointestinal tract showing ulcerative lesions. An antigen-capture ELISA is also available to detect the presence of BVDV antigen in blood or serum. The ELISA for BVDV antigen will detect viral infection and is widely used to diagnose persistently infected calves. Two samples taken 3-4 weeks apart will confirm a persistent infection. Immunohistochemistry will demonstrate the presence of antigen in fixed or frozen sections. Viral RNA may also be detected, using PCR for clinical specimens or in situ hybridisation on fresh or fixed tissues[40].
Genotype is generally determined by PCR with subsequent nucleic acid sequencing.
Pathology
In cases of mild, acute BVD, lesions are rarely seen. When disease is more severe, the lymph nodes may appear swollen, there may be erosions and ulcerations of the gastrointestinal tract tract and serosal surfaces of the viscera may show petechial and ecchymotic hemorrhages[40].
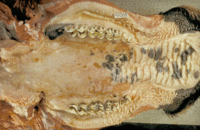
The pathology associated with mucosal disease is much more striking[31]. Oral, lingual and buccal erosions are observed, and buccal lesions often coalesce to form larger areas of necrosis and sloughed epithelium. Oesophageal lesions present similarly. The gastrointestinal tract often shows characteristic pathology, but post-mortem examination must be performed soon after death so that these are not masked by autolytic changes. In the rumen, ulceration is less common but, with congestion and oedema, may be seen along the pillars, and papillae can be reduced in size. Several discoid erosions of around 5mm in diameter appear in the abomasum, with hyperaemia of the surrounding mucosa and petechiation of the submucosa, particularly at the pylorus. Abomasal erosions occasionally enlarge and ulcerate. Oval erosions can be seen along the antimesenteric surface of the small intestine, overlying the lymphatic tissue of the Peyer's patches and measuring 2-5 centimetres in length. The erosions become larger and more numerous towards the terminal ileum, and the exposed surfaces varies in appearance. In more chronic lesions, food is seen to adhere to the underlying submucosa, and in acute disease the exposed surface is acutely congested and often haemorrhages into the gut lumen. In the large intestine, the mucosal folds may be thickened, giving the organ a striped appearance inwardly. Petechiation and erosions are occasionally seen along the folds, and the large intestinal contents are watery, dark and foul-smelling.
Treatment and Control
Acute BVDV infection is usually mild and does not require treatment, and treatment of more severe cases is symptomatic and supportive. There is no known treatment for mucosal disease and cases are euthanased on welfare grounds; recovery is most unlikely.
Control of BVD is practiced to a greater extent than treatment. The aim is eradication on individual farms but some countries, for example in Scandinavia, have achieved national eradication. There are several elements to control, including effective biosecurity, strategic testing, elimination of persistently infected animals, and vaccination strategies[40].
Biosecurity measures can include the usual hygiene precautions taken on farms, by visitors and during veterinary attention, as well as scrutiny of bought-in livestock and biologicals. Replacement cattle should be tested for persistent infection and quarantined on-farm in case of acute infections before entering the herd. If the resident herd is BVD-vaccinated, new animals should be brought up to date before joining the cohort. Embryo donors should be tested for persistent infection before transfer occurs, and purchase of in-calf heifers should be avoided as their offspring may be persistently infected. BVDV is shed in semen, so breeding bulls and semen for artificial insemination should be tested before coming into contact with cows.
A protocol for screening cattle herds for persistent infection should be implemented. Testing can be achieved by virus isolation or antigen-capture ELISA from serum or buffy coat cells, or by antigen detection in skin biopsies. The selected programme should be designed around the type and size of herd, financial limitations and the techniques available at the chosen diagnostic laboratory. Once identified, persistently infected animals should be culled as soon as possible.
Both modified live (not in the UK) and chemically-inactivated BVDV vaccines are available for use. Although cross-protection between strains and genotypes is generally good, antigenic diversity among challenge viruses may affect the efficacy of a given vaccine. Because BVDV is tropic for the foetus, modified live vaccines should not be used in pregnant animals. The virus is also immunosuppressive and so modified-live vaccination of animals showing signs of disease is not recommended. Maternally-derived antibody wanes at 3-6 months of age, and so to ensure that vaccination induces a protective immune response animals should be vaccinated (or re-vaccinated) by this age.
Links
- Royal Veterinary College BVD page
- Bovine Viral Diarrhoea (BVD): An Eradication Scheme for Scotland - Consultation Paper
| Bovine Viral Diarrhoea Virus Learning Resources | |
|---|---|
 Selection of relevant videos |
Bovine Viral Diarrhoea Virus (BVDV) - part 1 Bovine Viral Diarrhoea Virus (BVDV) - part 2 Bovine Viral Diarrhoea Virus (BVDV) - part 3 |
 Search for recent publications via CAB Abstract (CABI log in required) |
Bovine Viral Diarrhoea Virus publications |
 Full text articles available from CAB Abstract (CABI log in required) |
Bovine viral diarrhoea: an emerging disease of ruminants in India. Richa Sood; Intas Pharmaceuticals Ltd, Ahmedabad, India, Intas Polivet, 2007, 8, 1, pp 145-152, 37 ref. |
References
- ↑ Collett, M S et al (1988) Proteins encoded by bovine viral diarrhoea virus: The genomic organisation of a pestivirus. Virology, 165(1), 200-208.
- ↑ Meyers, G et al (1989) Molecular Cloning and nucleotide sequence of the genome of hog cholera virus. Virology, 171(2), 555-567.
- ↑ 3.0 3.1 3.2 3.3 3.4 Donis, R O(1995) Molecular biology of bovine viral diarrhea virus and its interactions with the host. The Veterinary Clinics of North America: Food Animal Practice 11(3), 393-424.
- ↑ 4.0 4.1 4.2 Dubovi, E J (1990) Molecular biology of bovine virus diarrhoea virus. Revue Scientifique et Technique, 9(1), 105-114.
- ↑ Van Gennip, H G P et al (2005) Dimerisation of glycoprotein Erns of classical swine fever virus is not essential for viral replication and infection. Archives of Virology, 150(1), 2271-2286.
- ↑ Tautz, N et al (1999) Establishment and Characterization of Cytopathogenic and Noncytopathogenic Pestivirus Replicons. Journal of Virology, 73(11), 9422–9432.
- ↑ 7.0 7.1 Harada, T et al (2000) E2-p7 Region of the Bovine Viral Diarrhea Virus Polyprotein: Processing and Functional Studies. Journal of Virology, 74(20), 9498–9506.
- ↑ Zhong, W et al (1998) Identification and Characterization of an RNA-Dependent RNA Polymerase Activity within the Nonstructural Protein 5B Region of Bovine Viral Diarrhea Virus. Journal of Virology, 72(11), 9365–9369.
- ↑ Paton, D J et al (1995) A proposed division of the pestivirus genus using monoclonal antibodies, supported by cross-neutralisation assays and genetic sequencing. Veterinary Research, 26, 82-109.
- ↑ Vilcek, S et al (2001) Bovine viral diarrhoea virus genotype 1 can be separated into at least eleven groups. Archives of Virology, 146, 99-115.
- ↑ 11.0 11.1 11.2 Carman, S et al (1998) Severe acute bovine viral diarrhea in Ontario, 1993-1995. Journal of Veterinary Diagnostic Investigation, 10, 27-35.
- ↑ Cortese, V S et al (1998) Clinical and immunologic responses of vaccinated and unvaccinated calves to infection with a virulent type-II isolate of bovine viral diarrhea virus. Journal of the American Veterinary Medical Association, 213, 1312-1319.
- ↑ Van Oirschot, J T et al (1999) Vaccination of cattle against bovine viral diarrhoea. Veterinary Microbiology, 64, 169-183.
- ↑ Deregt, D et al (2004) Attenuation of a virulent type 2 bovine viral diarrhea virus. Veterinary Microbiology, 100, 151-161.
- ↑ Corapi, W et al (1989) Severe Thrombocytopenia in Young Calves Experimentally Infected with Noncytopathic Bovine Viral Diarrhea Virus. Journal of Virology, 63(9), 3934-3943.
- ↑ Ahn, B C et al (2005) Biotype, Genotype, and Clinical Presentation Associated With Bovine Viral Diarrhea Virus (BVDV) Isolates From Cattle. International Journal of Applied Research in Veterinary Medicine, 3(4), 319-325.
- ↑ 17.0 17.1 17.2 Brownlie, J., Clarke, M. & Howard, C. (1984) Experimental production of fatal mucosal disease in cattle Veterinary Record, 114, 535-536
- ↑ Deregt, D and Loewen, K G (1995) Bovine viral diarrhea virus: Biotypes and disease. Canadian Veterinary Journal, 36, 371-378.
- ↑ Donis, R O and Dubovi, E J (1987) Differences in virus-induced polypeptides in cells infected by cytopathic and noncytopathic biotypes of bovine virus diarrhea-mucosal disease virus. Virology, 158, 168-173.
- ↑ Pocock, D H et al (1987) Variation in the intracellular polypeptide profiles from different isolates of bovine viral diarrhea virus. Archives of Virology, 94, 43-53.
- ↑ Magar, R et al (1988) Bovine viral diarrhea virus proteins: heterogeneity of cytopathogenic and noncytopathogenic strains and evidence of a 53K glycoprotein neutralization epitope. Veterinary Microbiology, 16, 303-314.
- ↑ Tautz, N et a; (1999) Establishment and Characterization of Cytopathogenic and Noncytopathogenic Pestivirus Replicons. Journal of Virology, 73(11), 9422–9432.
- ↑ Meyers, G and Thiel, H J (1996) Molecular characterization of pestiviruses. Advances in Virus Research, 47, 53-118.
- ↑ 24.0 24.1 Houe, H (1999) Epidemiological features and economical importance of bovine virus diarrhoea virus (BVDV) infections. Veterinary Microbiology, 64, 89-107.
- ↑ Kirkland, P D et al (1991) Replication of bovine viral diarrhoea virus in the bovine reproductive tract and excretion of virus in semen during acute and chronic infections. Veterinary Record, 128, 587–590.
- ↑ Gunn, H M (1993) Role of fomites and flies in the transmission of bovine viral diarrhoea virus. Veterinary Record, 132, 584-585.
- ↑ Lang-Ree, J R et al (1994) Transmission of bovine viral diarrhoea virus by rectal examination. Veterinary Record, 135, 412-413.
- ↑ Tarry, D W et al (1991) Transmission of bovine virus diarrhoea virus by blood feeding flies. Veterinary Record, 128(4), 82-84.
- ↑ Bruschke, C J M et al (1998) Distribution of bovine virus diarrhoea virus in tissues and white blood cells of cattle during acute infection. Veterinary Microbiology, 64, 23-32.
- ↑ Spagnuolo, M et al (1997) Bovine Viral Diarrhoea Virus Infection in Bone Marrow of Experimentally Infected Calves. Journal of Comparative Pathology, 116, 97-100.
- ↑ 31.0 31.1 31.2 31.3 31.4 31.5 31.6 31.7 Brownlie, J (1985) Clinical aspects of the bovine virus diarrhoea/ mucosal disease complex in cattle. In Practice, 7(6), 195-202.
- ↑ Ames, T R (1986) The causative agent of BVD: Its epidemiology and pathogenesis. Veterinarni Medicina, 81, 848-869.
- ↑ 33.0 33.1 Perdrizet, J A et al (1987) Bovine virus diarrhea – clinical syndromes in dairy herds. Cornell Veterinarian, 77, 46-74.
- ↑ Moerman, A et al(1994) Clinical consequences of a bovine virus diarrhoea in a dairy herd: A longitudinal study. Veterinary Quarterly, 16, 115-119.
- ↑ 35.0 35.1 35.2 35.3 Duffell, S J and Harkness, J W (1985) Bovine virus diarrhoea-mucosal disease infection in cattle. Veterinary Record, 117, 240-245.
- ↑ 36.0 36.1 36.2 36.3 36.4 Baker, J (1995) The Clinical Manifestations of Bovine Viral Diarrhea Infection. The Veterinary Clinics of North America: Food Animal Practice, 11(3), 425-445.
- ↑ Martin, S W and Bohac, J G (1986) The association between serologic titers in infectious bovine rhinotracheitis virus, bovine virus diarrhea virus, parainfluenza-3 virus, respiratory syncitial virus and the treatment for respiratory disease in Ontario feeder calves. Canadian Journal of Veterinary Research, 50, 351-358.
- ↑ Hibberd, R C, Turkington, A & Brownlie, J.(1993) Fatal bovine viral diarrhoea virus infection of adult cattle. Veterinary Record, 132, 227-228.
- ↑ Rebhun, W C et al (1989) Thrombocytopenia associated with bovine viral diarrhoea infection in cattle. Journal of Veterinary Internal Medicine, 3, 42-46.
- ↑ 40.0 40.1 40.2 40.3 40.4 Merck & Co (2008) The Merck Veterinary Manual (Eighth Edition), Merial.
- ↑ Carlsson, U et al (1989) Bovine virus diarrhoea virus: A cause of early pregnancy failure in the cow. Journal of Veterinary Medicine, 36, 15-23.
- ↑ Mc Gowan, M R et al (1993) Increased reproductive losses in cattle infected with bovine pestivirus aroung the time of insemination Veterinary Record, 133, 39-43.
- ↑ Bolin, S R et al (1985) Response of cattle persistently infected with noncytoparthic bovine viral diarrhea virus to vaccination for bovine viral diarrhea and subsequent challenge exposure who cytopathic bovine viral diarrhea virus. American Journal of Veterinary Research, 46, 2467-2470.
- ↑ Houe, H (1993) Survivorship of animals persistently infected with bovine virus diarrhoea virus (BVDV) Preventative Veterinary Medicine, 115, 275-283.
- ↑ Brownlie, J (1990) Pathogenesis of mucosal disease and molecular aspects of bovine virus diarrhoea virus Veterinary Microbiology, 23, 371-382.
- ↑ Brownlie, J (1990) Pathogenesis of mucosal disease and molecular aspects of bovine virus diarrhoea virus Veterinary Microbiology, 23, 371-382.
| This article has been expert reviewed by Prof Joe Brownlie DSc BVSc PhD FRCVS FRCPath DipECVP Date reviewed: July 8, 2011 |
Error in widget FBRecommend: unable to write file /var/www/wikivet.net/extensions/Widgets/compiled_templates/wrt6a29aaf4df3264_02367957 Error in widget google+: unable to write file /var/www/wikivet.net/extensions/Widgets/compiled_templates/wrt6a29aaf501c038_60670263 Error in widget TwitterTweet: unable to write file /var/www/wikivet.net/extensions/Widgets/compiled_templates/wrt6a29aaf50fa917_61896125
|
| WikiVet® Introduction - Help WikiVet - Report a Problem |