Aspergillosis
Also known as: Brooder Pneumonia in young birds
Introduction
Apergillosis is a disease of the respiratory system caused by several Aspergillus spp.. Aspergillus fumigatus is the most frequently encountered species in domestic animals but Aspergillus tereus and Aspergillus flavus have also been reported. Aspergillus is a ubiquitous saprophyte and is found worldwide. It is also a component of normal hair, skin and mucosal flora in both humans and animals. Commonly affected species include birds, dogs, cats, horses and cattle but the disease has been reported in many other wild and domestic species.
Pathogenesis
In dogs, spores are inhaled from the environment leading to fungal colonisation of the nasal cavities. Following their deposition in tissue and recognition by phagocytes, an inflammatory response is triggered. Haemolytic and dermonecrotic toxins as well as fungal protease and elastase are released leading to tissue damage.
In horses, the pathogenesis of guttural pouch mycosis is largely unknown but is thought to relate to damage to the mucosal layer of the pouches by trauma or infection. This enables opportunistic Aspergillus fungi to invade and colonise the damaged tissue.
Clinical Signs
Dogs
Aspergillosis is a common cause of nasal disease in dogs. Cases occur most commonly in young to middle-aged male dogs, but there is no apparent age or sex predilection. A higher prevalence of disease has been reported in doliocephalic breeds and outdoor/farm dogs. Clinical signs are those seen with any chronic nasal disease and include sneezing, unilateral or bilateral serosanguinous nasal discharge, ulceration of the nares, nasal pain and epistaxis. Neurological signs may be displayed if there is involvement of the cribriform plate.
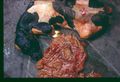
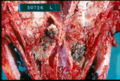
The disease is usually localised to the paranasal sinuses and nasal cavity but a disseminated form with granulomas and infarcts has been reported, particularly in German Shepherds. This form of disease often involves multiple organ systems including the spleen and kidneys. Clinical signs include lethargy, anorexia, urinary incontinence and haematuria. The vertebrae are frequently affected and osteomyelitis and discospondlylitis are common features. Dermatological signs of disseminated aspergillosis include abscesses, draining tracts, oral ulcers and cutaneous nodules.
Horses
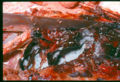
In the horse, Aspergillus most commonly affects the guttural pouches but infection may also lead to abortion, keratomycosis and rarely pulmonary aspergillosis. There is no age, sex or breed predisposition for guttural pouch mycosis and both left and right pouches are affected with equal frequency. Guttural pouch mycosis is characterised by spontaneous epistaxis (often in a resting horse) as a result of fungal erosion of the internal carotid artery. Other clinical signs include nasal discharge and dysphagia. Mycotic plaques are usually located on the caudodorsal aspect of the medial guttural pouch.
Cattle
Aspergillosis has a number of clinical manifestations in the cow including mastitis, placentitis, diarrhoea, ocular infection and mycotic pneumonia. Abortion in the second or third trimester of pregnancy has also been described. In the case of pulmonic disease, clinical signs may include pyrexia, cough, dyspnoea and tachypnoea but may be limited to vague signs such as weight loss or signs of mild respiratory disease. In aborting cattle, the foetus and placenta are retained and foetal lesions such as bronchopneumonia and dermatitis may be seen. Mastitic cows may display depression, weight loss and pyrexia with purulent mammary secretions and a hot, swollen udder.
Birds
Three forms of the disease have been reported in avian species; a diffuse infection of the air sacs; a diffuse pneumonic form and a nodular form involving the lungs. In chicks and poults the disease is known as 'brooder pneumonia' and may affect many birds in a flock. It is a very common disease, especially in pet birds due to the poor environments in which the birds are kept, combined with the anatomy of their respiratory system. Animals become infected due to inhalation of spores from contaminated feed or litter. These spores remain lodged in the respiratory system without causing clinical disease until the bird is stressed. Disease normally occurs in the syrinx or the air sacs. Clinical signs include dyspnoea, diarrhoea, listlessness, pyrexia, loss of appetite and loss of condition. Seizures and torticollis may occasionally occur if infection disseminates to the brain. Eggs may also be infected, with the fungal infection growing in the air cell of the membrane within the egg. Embryos may die before hatching or be weak and dyspnoeic after hatching. Occasionally, nervous system problems or diarrhoea are found in birds that survive to hatch.
Diagnosis
Dogs
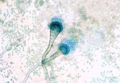
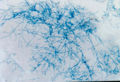
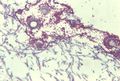
Radiology is often performed in the diagnostic work up of an animal with suspected Aspergillosis. It should always be performed prior to other procedures such as rhinoscopy and biopsy in order to prevent haemorrhage that may obscure subtle radiographic findings. Open-mouth ventro-dorsal views often reveal generalised radiolucency and lysis of the turbinate bones. Additionally, cytological examination of aspirates often reveals presence of fungal hyphae with granulomatous to suppurative inflammation and necrosis. Rhinoscopy may also be used to directly visualise the lesions, revealing characteristic white-green fungal plaques and destruction of the nasal turbinates. It also allows collection of material for fungal culture. This may be achieved using Sabouraud's dextrose agar in order to demonstrate the organism but should not be used as the sole means of diagnosis due to the ubiquitous nature of Aspergillus in the environment. White colonies form initially which turn dark green, flat and velvet-like in appearance. Serological findings such as immunoelectophoresis, ELISA and agar gel diffusion may provide additional diagnostic information.
Horses
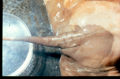
Diagnosis is obtained following endoscopic examination of the guttural pouches and observation of white-yellow-black mycotic plaques on the mucosal surface of the guttural pouches. Care must be taken whilst performing endoscopy due to the risk of dislodgement of a thrombus on the affected artery.
Birds
Endoscopy allows visualisation of the fungus and is the fastest and simplest way to obtain a tentative diagnosis. Culture of the lesions is required for definitive diagnosis. Post-mortem findings include bronchial or tracheal plugs, air cell plaques or small yellow nodules in the lungs. When Aspergillus infection is suspected in the embryo positive culture of lesions on the air cell membrane, lungs, air sacs or air passages is required for definitive diagnosis.
Treatment
Dogs
Several different treatment options are available.
Ketoconazole can be administered orally for 6-8 weeks. Itraconazole has also been used. This can be effective in up to 50% of cases.
Another non-invasive option includes infusion of enilconazole or clotrimazole into the external nares whilst occluding the nasopharynx. The animal is first placed in dorsal recumbency, then switched to ventral recumbency and any fluid is allowed to drain from the nose. This technique may not be effective and may lead to aspiration pneumonia or oesophagitis from leakage of the agent.
The treatment of choice is topical application of the anti-fungal agent Clotrimazole. It is administered for one hour under general anaesthetic via indwelling catheters placed in the frontal sinus. Several treatments may be required. For cases that are non-responsive to Clotrimazole, treatment with Enilconazole may be attempted but this is associated with a higher complication rate.
Horses
Trans-arterial coil embolisation under fluoroscopic guidance is performed in order to cause internal occlusion of the affected arteries. Following arterial occlusion the mycotic plaques usually resolve without necessitating further treatment.
Cattle
Antifungal agents are currently unlicensed and management of the disease usually relies on preventative measures such as ensuring clean bedding and good husbandry.
Birds
Birds should be treated with both anti-fungal drugs and F10 disinfectant, delivered via nebulisation. This treatment should continue for a minimum of 6 weeks up to a period of 6 months, therefore cost considerations may need to be discussed with the owners. Following the cessation of treatment, repeat endoscopy should be performed to ensure the disease has resolved. Aspergillosis of the syrinx may present as an emergency due to obstruction of the airway. This requires surgical removal of the fungal growths. An air sac tube may be placed to stabilise the bird prior to the planned surgery, and endoscopy should be performed on the lungs and air sacs to assess the spread of the fungus and provide a prognosis (poor with further spread) before surgery is performed. A low dust level, high quality feed and frequent F10 fogging can help to prevent the disease. There is currently no recommended treatment for eggs infected with aspergillosis, therefore efforts should be concentrated on prevention. This is especially important in air-forced incubators, where aspergillosis can be a serious problem. Incubators and hatchers should be cleaned and fumigated between batches of eggs, and an 'all in, all out' system should be in operation. UV sterilization of eggs prior to incubation and sanitizing dips or washes may be used to reduce the chances of infection. Eggs should be examined for cracks and weighed at regular intervals, and dead eggs should be removed immediately as infection of viable eggs by dead Aspergillus-infected eggs can occur. Also, eggs from damp or wet nests should not be incubated.
| Aspergillosis Learning Resources | |
|---|---|
To reach the Vetstream content, please select |
Canis, Felis, Lapis or Equis |
 Test your knowledge using flashcard type questions |
Aspergillosis Flashcards |
 Search for recent publications via CAB Abstract (CABI log in required) |
Aspergillosis in cats publications Aspergillosis in horses publications Aspergillosis in cattle publications Aspergillosis in birds publications Aspergillosis in sheep publications Aspergillosis in goats publications Aspergillosis in pigs publications |
 Full text articles available from CAB Abstract (CABI log in required) |
Aspergillosis: a sapromycotic zoonosis. Mahendra Pal; Pratibha Dave; Intas Pharmaceuticals Ltd, Ahmedabad, India, Intas Polivet, 2006, 7, 2, pp 421-428, 44 ref. |
References
- Barr, S. C., Bowman, D. D. (2006) The 5-minute Veterinary Consult Clinical Companion: canine and feline infectious diseases and parasitology Wiley-Blackwell
- Brown, SA & Rosenthal KL (1997) Self-Assessment Colour Review Small Mammals Manson Publishing Ltd
- Carter, G. R., Wise, D. J. (2004) Essentials of Veterinary Bacteriology and Mycology Wiley-Blackwell
- Ettinger, S. J. (2000) Pocket Companion to Textbook of Veterinary Internal Medicine Elsevier Health Sciences
- Muller, G. H., Scott, D. W., Kirk, R. W., Miller, W. H., Griffin, C. E. (2001) Muller and Kirk's Small Animal Dermatology Elsevier Health Sciences
- Stanford, M (2009) Respiratory Disease in Birds RVC Intergrated BVetMed Course, Royal Veterinary College
| This article has been peer reviewed but is awaiting expert review. If you would like to help with this, please see more information about expert reviewing. |
Error in widget FBRecommend: unable to write file /var/www/wikivet.net/extensions/Widgets/compiled_templates/wrt69fbb93fddda39_01454378 Error in widget google+: unable to write file /var/www/wikivet.net/extensions/Widgets/compiled_templates/wrt69fbb93febe162_29724435 Error in widget TwitterTweet: unable to write file /var/www/wikivet.net/extensions/Widgets/compiled_templates/wrt69fbb9400e7ff3_49462490
|
| WikiVet® Introduction - Help WikiVet - Report a Problem |