Innate Immunity Cellular Responses
Introduction
Pathogens can invade the body if a breach occurs in the barriers formed by the skin and mucus membranes, for example a wound, they must be detected and destroyed by cellular and humoral means.
The cells involved in the cellular response to a wound are:
- Tissue mast cells and macrophages that initially phagocytose and detect bacteria or fungi
- The blood granulocytes, or polymorphonuclear (PMN: multi-lobed nuclei) cells
- The neutrophils are the most abundant as they are the primary cells that phagocytose bacteria, and the larger fungi
- The eosinophils and basophils / mast cells are only needed in rare circumstances as they are for killing parasites by the release of granules (exocytosis).
- Blood monocytes: phagocytose bacteria
The main role of the innate cellular response is to delay systemic infection until the adaptive response can back it up with a more specific attack.
Macrophages
The innate cellular response begins with recognition and phagocytosis by macrophages that precide in the tissue where the wound occurs. The populations of distinct, tissue-specific macrophages that are present are the:
- Alveolar macrophages (lung)
- Tissue histiocytes
- Glomerular macrophages (kidney)
- Hepatic Küpffer cells (liver)
- CNS microglia (central nervous system: brain and spinal cord)
- Sinus-lining macrophages of the lymph nodes and spleen
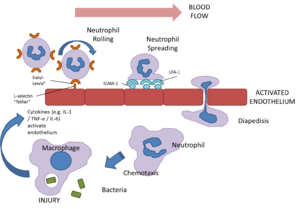
When phagocytosis of the bacteria or fungi by the tissue macrophages occurs, they release a number of cytokines that have many effects. The first being the activation of the endothelium that results in the expression of adhesion molecules that allow leukocytes, in particular the neutrophils to bind to the endothelium from the bloodstream and the subsequent extravasation (movement out of the bloodstream). Once the leukocytes are out of the bloodstream, the cytokines released by the tissue macrophages act as chemoattractants that enable the chemotaxis of the leukocytes to the site of infection.
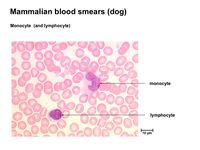
The Monocytes are immature macrophages that are circulating phagocytes. They circulate for 6-8 hours and function as phagocytes within the blood and as newly migrated cells in tissues. Their primary function, however, is to replace the various tissue macrophage populations at the latter stages of the immune response.
Neutrophils
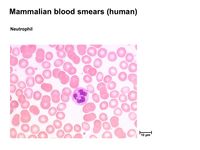
Neutrophils are the principal, highly active phagocytes in the blood and comprise 30-70% of all white blood cells depending on species. Their main function is to kill and digest microbes in a similar way as macrophages. They also have another function of extracellular bacterial killing by disrupting bacterial membranes by the secretion of small antibacterial peptides, for example defensins and bactenecins.
Neutrophils also produce vasoactive peptides, for example, histamine and bradykinin which, as their name suggests, activate the endothelium to become more "leaky" causing a great increase in extravasation of blood granulocytes and monocytes, and the diffusion of plasma proteins to the site of infection. These peptides, released from other cells as well as neutrophils, are responsible for the classical signs of inflammation: redness (rubor), heat (calor), swelling (tumour), and pain (dolor), often accompanied by loss of function. Neutrophil activation in an inflammatory lesion also results in the release of prostaglandins which are responsible for vasoactive changes and for pain (N.B. These are reduced with cyclo-oxygenase (COX) inhibition for example with the NSAIDs (non-steriodal anti-inflammatory drugs) aspirin and ibuprofen).
It is for the reasons above that neutrophils have been described as the archetypal cell associated with acute inflammation.
To move the neutrophils from the blood to the sites of inflammation a system of various chemoattractants exist. These include complement components, in particular C3a and C5a; cytokine production, in particular the chemokine class of cytokines, for example CXCL8 (IL-8); and the activation of the endothelium of post-capillary venules.
Their removal from the site after the removal of infection is an important step in the resolution of the lesion. Under physiological conditions the neutrophils undergo a process called apoptosis (or Programmed Cell Death), and are then cleared by tissue macrophages (see above). However, if there are too few macrophages to clear the cells, or the infection is resulting in the death of the neutrophils, they undergo a process called necrosis. When these neutrophils that have undergone necrosis accumulate at the site of infection it forms pus.
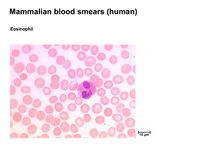
Eosinophils
Eosinophils are less common than neutrophils, with eosinophils making up less than 5% of the leukocytes in normal blood. They are still described as granulocytes but they are not phagocytic.
It has been found that eosinophil numbers are increased slightly during the resolution phase of inflammation, but many-fold in parasite-infected animals. It is therefore the presence of a large proportion of eosinophils in a blood smear that is highly indicative of parasitaemia.
The eosinophils mainly function by targeting the surface of parasites by means of a specific antibody or complement component. Once the parasite has been recognised the eosinophils release, through a process called exocytosis (the fusion of granules to the plasma membrane), a large range of toxic molecules that break down the parasite integument.
Intrestingly, eosinophils are also prominent in allergic (anaphylactic) reactions.
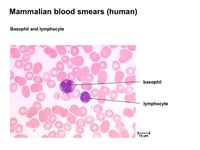
Basophils / Mast Cells
The basophils and the mast cells are principally localised at epithelial surfaces with very small numbers present in blood, making up less than 0.5% of circulating leukocytes.
These two cell types have two principal functions:
The first is the induction of acute inflammation. They do this through the production of cytokines by the mast cells in response to trauma and/or bacterial infection which then induces a classical acute inflammatory response.
The second is to respond to parasite infection. They do this specifically by interacting with the Fc region of the IgE class of antibody. When the IgE then comes into contact with antigen, Fc receptors on the surface of the mast cells and basophils can interact with the antibody which causes the mast cells and basophils to degranulate. This causes the release of enzymes and vasoactive substances that can result in a high level of mucus secretion and smooth muscle contraction. As the granules contain a much greater concentration of these peptides, the degranulation of mast cells and basophils creates a much more powerful response than that by neutrophils alone (it is for this reason that they can cause Type I hypersensitivity reactions). These cells also produce factors that influence local host cell physiology
and various mediators that increase the ratio of phagocyte to microbe (in particular cytokines).
| This article has been expert reviewed by Dr Robert J Francis BSc(Hons) PhD Date reviewed: May 3, 2012 |
| Originally funded by the RVC Jim Bee Award 2007 |