Varroa destructor
| Varroa destructor | |
|---|---|
| Phylum | Arthropoda |
| Class | Arachnida |
| Order | Mesostigmata |
| Family | Varroidae |
| Genus | Varroa |
| Species | V.destructor |
Also known as: Honeybee mite — Varroa jacobsoni
Causes: Varroatosis — Varoosis — Colony Collapse Syndrome
Introduction
The Varroa destructor mite is an external mite of honey bees. It causes varroatosis, which is no longer a notifiable disease in the UK.
Identification
The mite is oval in shape and 1-1.5mm in length. It is reddish-brown in colour and has 8 legs.
Life cycle
The mite has a 10 day life cycle. Eggs are laid in the hive and develop with the brood cells into mites.
Pathogenesis
The mites are blood-sucking, feeding on the bees and leaving open wounds. This weakens adult bees and damages growing larval bees, resulting in deformities.
Description
Varroosis is the clinical syndrome caused by the infestation of colonies of honey bees (Apis mellifera) by the mite Varroa destructor, formerly Varroa jacobsoni. Varroosis is a complex syndrome that may result in so-called Colony Collapse Syndrome, failure to survive hibernation or complete absconcion from the hive. V. destructor was traditionally a parasite of the Asian honey bee (Apis cerana) which is much more resistant to infestations but it began to infest European bees around fifty years ago and it has spread to all parts of the World except for Australia[1]. So far, only two mitochondrial haplotypes of V. destructor (Korea and Japan) have been found to have made the transition from A. cerana to A. mellifera[2]. The mite is able to infect colonies of Africanised ('killer') honey bees but these do not appear to show colony collapse syndrome[3].
Pathophysiology
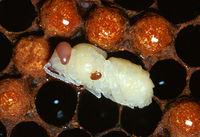
The fertilised adult female mite enters a random cell[4] just before it is sealed and initially feeds on the brood food. When the bee egg is laid and the cell sealed, the mite bites through the integument of the larva (usually towards its caudal end in the second sternite of the abdomen) and begins to suck its haemolymph. The same wounds are used by all of the mites in the cell and they rapidly become infected with bacteria[5]. The wounds are generally sealed with scar tissue after the first imaginal moult[6]. She lays eggs after around 72 hours and she and her offspring continue to feed on the developing bee. In heavy infestations, several female mites may enter a single cell. The next generation of mite offspring are produced in the cell[7] and these individuals continue to inter-breed after 6-7 days of development. A female mite first produces a male offspring then 5-6 female offspring, giving the mite a high biotic potential. The sequence of sexes seems to be dependent on a signal from the developing brood as female mites that are moved to a new cell after laying the male egg start an entirely new breeding cycle, apparently caused by the younger age of the new brood[8]. When the developed bee breaks out of the cell, it carries adult female mites into the hive but immature stages and male mites are left to die. The female must therefore be fertilised before she leaves the males behind, one reason why the infestation is sometimes romantically called Amazon disease. The female mites show a predilection for the cells of drones[9], probably because these brood take longer to develop and therefore allow a further generation of mites to develop in the cell. However, unlike Varroa infestations of the Asian honey bee, it is able to develop in any type of cell. Developing larvae and pupae that suffer mite infestations often die or appear abnormal at emergence, often being smaller, weaker and having incomplete wings. Infested drones are unable to fly as far as uninfested bees and they have a lower sperm production[10] and it has been shown that worker bees that were infested with V. destructor as pupae are not able to make physiological changes needed for survival over winter[11].
Africanised honey bee colonies are resistant to infection by V. destructor, a finding that may be related to the fact that they build narrower cells[12].
The female mites preferentially attach to worker bees of the nursery group, probably due to the differential expression of alkenes on the surfaces of these and the older pollen gatherers[13] and due to the production of different pheromones by different groups of bees[14]. The mites also express cuticular hydrocarbons that mimic those of the bees they parasitise and the pattern of expression of these hydrocarbons changes according to the stage of development of the bee[15]. The mites suck haemolymph from the adult bees and also transfer numerous pathogenic viruses between individual bees. The feeding process may also make these worker bees more susceptible to the diseases caused by these viruses after they are introduced. The female mites have a variable lifespan depending on the time of year. In winter when the bees are hibernating, they may survive of up to five months but in summer, they generally live for 3-4 weeks. The mite reproductive ratio falls during winter as a greater proportion of male mites die before reaching maturity, reducing the number of female mites that become fertilised[16].
Epidemiology
Mites are transferred between colonies by worker bees in areas of high hive density by robbing (where worker bees steal honey from other hives), drifting (where worker bees become attached to another colony) or swarming (where unfertilised queens mix with drones from several colonies to become mated). Since its first reported infestation in the UK in 1992[17], V. destructor has now spread across the whole country except for the Isles of Scilly, the Isle of Man and some Scottish islands. Mites can also be spread iatrogenically as they survive on fomites for up to 72 hours and this is thought to be the major route of transmission.
Diagnosis
V. destructor adult mites are visible to the naked eye but they should be differentiated from several other mite species that affect honey bees (see below). They may be seen attached to worker bees or within cells from which workers have recently emerged. Live mites may drop from worker bees and be seen in the bottom of a hive.
Clinical Signs
Individual Mites
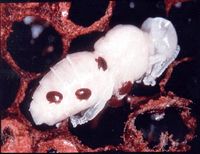
Bees that were infested by the mite during development may be small and weak, they may have wing deformities and they may have a reduced lifespan compared to normal bees. Dead brood may be seen within cells where they died as a result of the infestation.
Colony Collapse
If the infestation is severe, so many brood are affected that dying workers are not replaced at the normal rate, reducing the size of the colony. As the worker population shrinks, the normal functions of the hive cannot be maintained and the colony disintegrates, a phenomenon described as colony collapse syndrome. In such heavy infestations, mites should be evident on worker bees and multiple female mites may enter each worker or drone cell. As workers fail to emerge, a speckled pattern begins to form on the comb as some cells are empty and others remain capped. Other workers usually remove these dead brood but these tasks are not completed in collapsing colonies. In an unsupervised hive, an infested colony can collapse within weeks and the dispersal of its remaining bees can spread the mite in the local area.
Infested colonies are less able to make preparations for hibernation, including storage of honey. These colonies may therefore not survive the winter and empty hives may be found on inspection in the spring.
Estimation of the Infestation
An infestation of 1,000 mites is considered to be the maximum tolerable level in colonies in the UK before the colony is in real danger of collapse. Ideally, each hive should be checked four times in the season to estimate the level of the infestation using one of the following methods:
- Mite drop involves assessing the number of V. destructor mites visible on the floor of the hive as a guide to the number affecting the bees of the colony. The likelihood of colony collapse can be predicted from the number of mites that drop from the colony each day at different times through the year.
- Uncapping of drone cells allows direct evaluation of the level of infestation in the colony. At least 100 cells should be uncapped to determine the percentage of cells that are infested and to determine whether multiple mites have entered each cell (a sign of heavy infestation). An infestation in which more than 5-10% of cells contain mites is considered to be severe and the colony would be at risk of collapse.
Differential Diagnosis
Several other mites infest honey bees or may be found within a hive and V. destructor must be differentiated from these.
| Differential | Significance | Distinguishing features | Image |
| V. destructor | As above. | The honey bee mite has an oval body with all 8 legs on one side of the body. | |
| The bee louse Braula coeca | The louse is insignificant as a cause of disease | The body has a more rounded shape (compared to the marked oval shape of V. destructor) and its 6 legs extend outside the margins of the body on either side. | |
| Tropilaelaps spp. | This mite is a Notifiable exotic parasite that may cause severe disease in honey bee colonies. | The mites are smaller than V. destructor and have 6 legs which are visible on either side of the mite. | |
| Melittiphis alvearius | These mites prey on other mites within the hive and cause no harm to bees. | The mites are much smaller than V. destructor but have a similar oval shape. |
Treatment and Control
Hang accaricaidal strips between combs in the hive, after autumn 'honey flow'. If the outbreak is isolated, destroy all colonies involved in this outbreak to try and prevent further spread. However, if the outbreak is widespread, monitor mite numbers and treat with acaracide.
| Varroa destructor Learning Resources | |
|---|---|
 Test your knowledge using flashcard type questions |
Mites Flashcards |
 Search for recent publications via CAB Abstract (CABI log in required) |
Varroa destructor publications |
References
- ↑ Stevenson MA, Benard H, Bolger P, Morris RS. Spatial epidemiology of the Asian honey bee mite (Varroa destructor) in the North Island of New Zealand. Prev Vet Med. 2005 Oct 12;71(3-4):241-52. Epub 2005 Sep 6.
- ↑ Solignac M, Cornuet JM, Vautrin D, Le Conte Y, Anderson D, Evans J, Cros-Arteil S, Navajas M. The invasive Korea and Japan types of Varroa destructor, ectoparasitic mites of the Western honeybee (Apis mellifera), are two partly isolated clones. Proc Biol Sci. 2005 Feb 22;272(1561):411-9.
- ↑ Medina LM, Martin SJ, Espinosa-Montaño L, Ratnieks FL. Reproduction of Varroa destructor in worker brood of Africanized honey bees (Apis mellifera). Exp Appl Acarol. 2002;27(1-2):79-88
- ↑ Salvy M, Capowiez Y, Le Conte Y, Clement J. Does the spatial distribution of the parasitic mite varroa jacobsoni oud. (Mesostigmata: varroidae) in worker brood of honey bee apis mellifera L. (Hymenoptera: apidae) rely on an aggregative process? Naturwissenschaften. 1999 Nov;86(11):540-3.
- ↑ Kanbar G, Engels W. Communal use of integumental wounds in honey bee (Apis mellifera) pupae multiply infested by the ectoparasitic mite Varroa destructor. Genet Mol Res. 2005 Sep 30;4(3):465-72.
- ↑ Kanbar G, Engels W. Ultrastructure and bacterial infection of wounds in honey bee ( Apis mellifera) pupae punctured by Varroa mites. Parasitol Res. 2003 Aug;90(5):349-54. Epub 2003 Mar 27.
- ↑ Akimov IA, Yastrebtsov AV. Peculiarities in ontogenesis and reproductive cycle of the mite Varroa jacobsoni (Parasitiformes, Varroidae) and its relation to the host Apis mellifera. Wiad Parazytol. 1991;37(1):99-102.
- ↑ Garrido C, Rosenkranz P. The reproductive program of female Varroa destructor mites is triggered by its host, Apis mellifera. Exp Appl Acarol. 2003;31(3-4):269-73.
- ↑ Le Conte Y, Arnold G, Trouiller J, Masson C, Chappe B, Ourisson G. Attraction of the parasitic mite varroa to the drone larvae of honey bees by simple aliphatic esters. Science. 1989 Aug 11;245(4918):638-9.
- ↑ Duay P, De Jong D, Engels W. Decreased flight performance and sperm production in drones of the honey bee (Apis mellifera) slightly infested by Varroa destructor mites during pupal development. Genet Mol Res. 2002 Sep 30;1(3):227-32
- ↑ Amdam GV, Hartfelder K, Norberg K, Hagen A, Omholt SW. Altered physiology in worker honey bees (Hymenoptera: Apidae) infested with the mite Varroa destructor (Acari: Varroidae): a factor in colony loss during overwintering? J Econ Entomol. 2004 Jun;97(3):741-7.
- ↑ Piccirillo GA, De Jong D. The influence of brood comb cell size on the reproductive behavior of the ectoparasitic mite Varroa destructor in Africanized honey bee colonies. Genet Mol Res. 2003 Mar 31;2(1):36-42.
- ↑ Del Piccolo F, Nazzi F, Della Vedova G, Milani N. Selection of Apis mellifera workers by the parasitic mite Varroa destructor using host cuticular hydrocarbons. Parasitology. 2010 May;137(6):967-73. Epub 2010 Feb 15.
- ↑ Pernal SF, Baird DS, Birmingham AL, Higo HA, Slessor KN, Winston ML. Semiochemicals influencing the host-finding behaviour of Varroa destructor. Exp Appl Acarol. 2005;37(1-2):1-26.
- ↑ Martin C, Salvy M, Provost E, Bagnères A, Roux M, Crauser D, Clement J, Le Conte Y. Variations in chemical mimicry by the ectoparasitic mite Varroa jacobsoni according to the developmental stage of the host honey-bee Apis mellifera. Insect Biochem Mol Biol. 2001 Mar 15;31(4-5):365-79.
- ↑ Martin SJ. Varroa destructor reproduction during the winter in Apis mellifera colonies in UK. Exp Appl Acarol. 2001;25(4):321-5.
- ↑ Rendall GF. Varroa jacobsoni found in the UK. Vet Rec. 1992 Apr 18;130(16):359.
Blood, D.C. and Studdert, V. P. (1999) Saunders Comprehensive Veterinary Dictionary (2nd Edition) Elsevier Science
Fox, M and Jacobs, D. (2007) Parasitology Study Guide Part 1: Ectoparasites Royal Veterinary College
| This article has been peer reviewed but is awaiting expert review. If you would like to help with this, please see more information about expert reviewing. |
Error in widget FBRecommend: unable to write file /var/www/wikivet.net/extensions/Widgets/compiled_templates/wrt6a14d1b79c91b7_63996240 Error in widget google+: unable to write file /var/www/wikivet.net/extensions/Widgets/compiled_templates/wrt6a14d1b7a121e2_96714571 Error in widget TwitterTweet: unable to write file /var/www/wikivet.net/extensions/Widgets/compiled_templates/wrt6a14d1b7a589c0_55740967
|
| WikiVet® Introduction - Help WikiVet - Report a Problem |