Pneumonia - Introduction and Classification
- Pneumonia
- Inflammation that takes place in the alveoli and their walls
- Can be grouped either according to
- Nature of the inflammatory process
- Aetiological agent
- Pattern of the lesion
- Patterns of lung inflammation vary depending on the aetiology, route and method of exposure and multiple host factors including age, general health, and immune status
- There are five general patterns of pneumonia:
Bronchopneumonia
Bronchointerstitial Pneumonia
Lobar Pneumonia
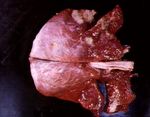
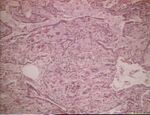
Interstitial pneumonia
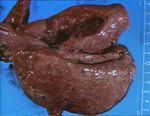
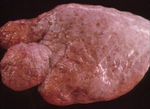
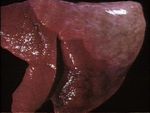
- Lung inflammation that occurs primarily in alveolar walls rather than in alveolar spaces
- Can be widely distributed but is often most severe in caudal lung lobes
- Diffuse or patchy damage to alveolar septa, may be acute or chronic
- Lesions in most cases result from blood-borne insult and are more likely to involve the dorsocaudal regions, although the damage is often diffuse
- No concentrated inflammatory response in the airway, only secondary to damage of alveolar walls
- As part of systemic infection
- Canine distemper (in alveolar macrophages as inclusions)
- Salmonellosis
- Toxoplasmosis (in alveolar wall)
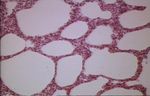
- Acute interstitial pneumonia is characterised by exudation into the alveolar lumen and in cattle is associated with interstitial emphysema
Examples:
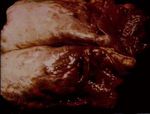
Acute bovine pulmonary emphysema and oedema (ABPEE)
- Synonym: fog fever
- Usually seen in adult beef cattle in the autumn as an outbreak
- Associated with a change in pasture (from dry to lush, green)
- Severe respiratory distress with laboured breathing and grunting on expiration
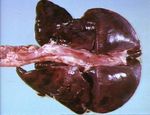
- Can result in diffuse fibrosing alveolitis
- Gross appearance is that of enlarged wet lungs, the interlobular septa are markedly widenedith oedema and emphysema
- Underlying pathogenesis is ingestion of L-tryptophan in the pasture which is metabolised to 3-methylindole -> bloodstream -> lungs -> metabolised into a compound toxic to Type 1 pneumonocytes and non-ciliated bronchiolar epithelium
- Their loss allows massive flooding of the alveoli with a protein-rich fluid
- The sequence of events in the lung is as follows:
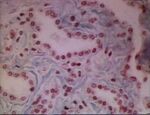
- Alveolar flooding with a protein-rich fluid due to the necrosis of Type 1 epithelium
- As the incoming air dries this fluid, fibrinous 'hyaline membranes' form
- Proliferation of the more resistant cuboidal Type 2 epithelium which line the alveoli, called 'epithelialisation'
- Then either there is
- Digestion of the hyaline membranes by macrophages which when completed, allows some of the proliferated Type 2 epithelium to differentiate into Type 1 and reconstitute the functional respiratory unit - a normal alveolus
- Or
- Organisation of the fibrin into fibrous tissue in the lumen - destroying the alveolus or proliferation of fibrous tissue in the alveolar wall with retention of the epithelialised appearance to the alveolus
- Sequel
- Residual scarring if animal survives initial onslaught
- In chronic exposures there may be extensive fibroplasia
- Ingestion of sweet potatoes ifested with Fusarium solani mould or pasture contaminated with stinkweed or purple mint, rapeseed and kale also cause pulmonary oedema, emphysema and interstitial pneumonia
Paraquat poisoning
- In dogs and cats
- Free radical release causes damage to air-blood barrier
- Depending on the dose, the lesions range from acute lesions (oedema, haemorrhage, hyaline membranes) -> chronic (fibroplasia of alveolar septae, replacement of alveolar cells with type II cells)
- Additional extrapulmonary lesions to note following paraquat intoxication are necrosis of the adrenal zona glomerulosa and renal tubular epithelium
Diffuse alveolitis
- Chronic disease of adult cattle occuring sporadically
- Probably caused by repeated subclinical incidents of fog fever or farmer's lung
- Farmer's Lung
- Extrinsic allergic alveolitis
- Hypersensitivity of ingested or inhaled moulds
- May occur as an outbreak or sporadically in adult cattle
- Hypersensitivity diseases often cause an lymphocytic interstitial pneumonia
- Chronic interstitial pneumonia progresses to fibrosis
- Sometimes called pneumonitis
Embolic pneumonia
- Lung inflammation caused by haematogenous spread of infections into the lung
- No orientation around airways and can be in any lung region but most often affects caudal lobes
- The inflammation is oriented around blood vessels, usually venules or alveolar capillaries in which septic emboli localise
Granulomatous pneumonia
- Variable number and distribution of caseous or non-caseous granulomas throughout the lung
- Must be distinguished from neoplasms
- Calcification may occur
- Caused by agents resistant to phagocytosis, mostly fungi, Mycobacterium bovis, Rhodococcus equi, foreign material, FIP
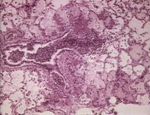
- Micro: centre of necrotic tissue surrounded by macrophages, connective tissue and lymphocytes
Verminous pneumonia
- Lesions in the lung may be due to:
- Parasites passing through the lung as part of their migration e.g. Ascaris suum in the pig, aberrant migration in the case of flukes;
- Parasites for which the lung is the target tissue e.g. Dictyocaulus viviparus
- Lesions can vary from interstitial to chronic bronchitis to granulomatous pneumonia
- May be infiltrate of eosinophils in pulmonary interstitium and bronchoalveolar spaces
- Atelectasis and emphysema may result due to obstruction
Stages of pneumonia
- Regardless of the pattern, all pneumonias pass through three stages:
- Exudative phase
- In this initial stage inflammatory exudate pours into alveolar spaces and alveolar capillaries are congested
- Type I alveolar epithelial cells are highly sensitive to injury and cannot proliferate in response to injury
- Necrosis and sloughing of injured type I cells, denuding alveolar spaces of lining epithelium
- Neutrophils begin to enter alveolar spaces distended with inflammatory oedema
- Proliferative phase
- Type II alveolar cells (less sensitive to and can proliferate in response to injury) begin to proliferate within 24 hours and eventually line the alveolar walls denuded of type I cells ***By 6 days cuboidal type II cells can completely line the alveoli
- Proliferation of type II cells marks the shift from the exudative to the proliferative stage of pneumonia, also heralded by decreased blood flow in alveolar capillaries
- Because the original squamous type I cells have been replaced by cuboidal type II cells, the microscopic appearance of pneumonic lungs at about 1 week has been described as “alveolar epithelialization”, “alveolar adenomatosis”, or “bronchiolisation of alveoli”
- Repair phase
- Resolution of pneumonia is accomplished by transformation of type II cells to type I cells
- Exudative phase
Pulmonary abscesses
- Commonly found post mortem
- Can be a consequence of:
- Septic emboli lodging in the pulmonary vessels
- Extension from severe focal suppurative bronchopneumonia
- Aspiration of foreign material (see aspiration pneumonia)
- Direct penetration
- The pattern of abscessation can suggest the aetiology i.e. multiple widespread abscesses suggest a haematogenous origin; isolated cranioventral abscesses usually arise from suppurative pneumonia.
Infectious causes of pneumonia
Other forms of pneumonia
Aspiration pneumonia
- Response of the lungs to aspirated foreign material will depend on the nature of the material (e.g. food), the bacterial load and the distribution of the material within the lungs
- Mild bronchopneumonia can develop into a severe necrotising pneumonia and in very severe cases can progress to gangrenous pneumonia (below)
- In Cattle
- Associated with poor husbandry
- Regurgitated ruminal content
- Cranio-ventral distribution
- Respiratory insufficiency secondary to congenital cardiac disease
- In Horses
- Most commonly in right ventral lung lobe (most rostral secondary bronchus leads to right accessory lobe)
- Risk factors:
- Oesophageal obstruction
- Spontaneous reflux (GI obstruction, equine grass sickness)
- Dysphagia
- Iatrogenic
- Nasogastric tube in the wrong place
- In Dogs
- Most commonly vomited/regurgitated material or contrast medium introduced into airways
- In severe acute cases may die from septic shock
- In chronic cases -> bronchopneumonia
- Common sequel to myasthenia gravis, megaoesophagus or persistent right aortic arch
Gangrenous pneumonia
- Can occur following severe infections in which tissue becomes necrotic and is then invaded by putrefactive saprophytes, however this is rare.
- The usual cause is administration of medicines (intended for the oesophagus!) or as a sequel to aspiration pneumonia
- The dead tissue undergoes liquefactive necrosis forming a cavity which is surrounded by intense hyperaemia and inflammation
Lipid pneumonia
- Associated with inhalation of oil, paraffin, etc.
- Reaction dominated by macrophages which fill the alveoli and interstitial thickening (mononuclear cells and fibrosis)
- Tends to acumulate in ventral regions bilterally
- Occurs subclinically in cats, sometimes dogs, unrelated to aspiration
- Gross lesion:
- Multifocal, firm, white nodules
- Microscopic lesions:
- Macrophages full of lipid forming foam within alveoli
- Interstitial lymphocyte and plasma cell infiltration, fibrosis
Uraemic pneumonia
- Severe uraemia causes increased permeability of the blood-air barrier and therefore can cause pulmonary oedema
- In addition to the oedema, there may also be degeneration and calcification of smooth muscle and connective tissue fibres
- Lungs do not collapse on opening the thorax in severe cases
Foetal pneumonia
- Especially foals and farm species
- Due to aspiration of amniotic fluid contaminated with meconium and bacteria
- Secondary to fetal distress
- Microscopic lesions of bronchopneumonia
- Involves all lobes (versus postnatal bronchopneumonia)
- Haematogenous spread
- Causes interstitial pneumonia
- Often caused by Listeria monocytogenes, Salmonella spp. or Chlamydia psittaci
- In viral abortions
- Cause bronchointerstitial pneumonia
- E.g.: IBR, PI-3 and equine viral rhinopneumonitis